 
The five most common were leucocytes decreased (65 percent), lymphocytes decreased (62 percent), platelet count decreased (53 percent), diarrhoea (47 percent) and rash (45 percent). 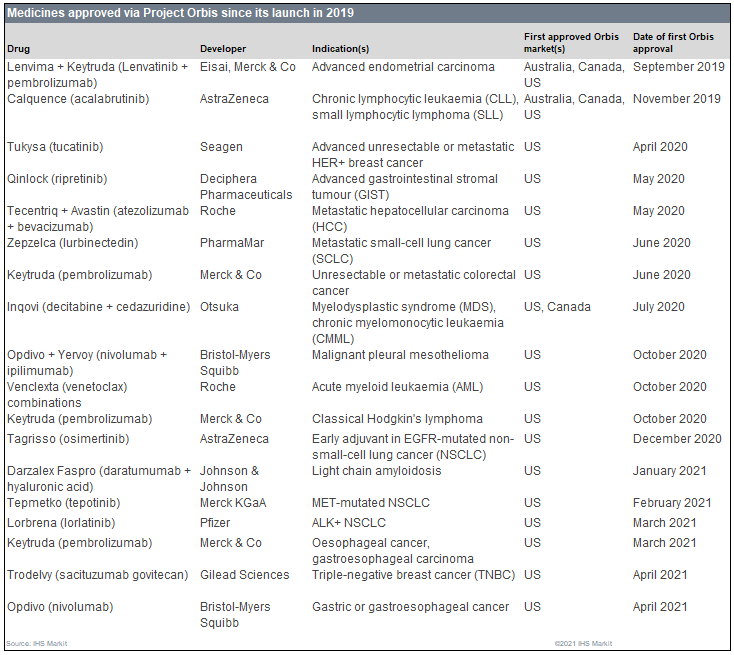
There are numerous adverse events associated with osimertinib however, in ADAURA and two accompanying trials with very few cases being worse than Grade 3. Moreover, at the two-year follow up, 89 percent of patients with stage IB-IIIA disease treated with osimertinib after surgery remained alive and disease-free versus 52 percent on placebo. Additionally, when looking at the broader group of patients, from stage IB to IIIA, treatment with osimertinib reduced the relative risk of disease recurrence or death by 80 percent, compared to placebo, after 24 months of treatment. The approval was based on the results of the ADAURA Phase III trial, in which the relative risk of disease recurrence or death for patients with stage II-IIIA EGFR mutation-positive NSCLC treated with osimertinib as an adjuvant treatment after surgery was reduced by 83 percent compared to placebo. Prior to the UK joining, the scheme has already given the green light to many life-saving treatments for patients suffering from conditions such as breast cancer, lung cancer, liver cancer, endometrial cancer and chronic lymphocytic leukaemia. The project is co-ordinated by the US Food and Drug Administration (FDA) and its other participants include Canada, Australia, Switzerland, Singapore and Brazil. Project Orbis is an innovative programme allowing partners from various countries to review and approve applications for promising cancer treatments quickly and efficiently. This authorisation is the first issued by the regulator under Project Orbis, having only joined the scheme in January 2021, following the UK’s exit from the European Union. According to the MHRA, the extended licence offers a novel treatment option for early-stage NSCLC patients, an area of significant unmet need. 
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) has authorised a licence extension for AstraZeneca’s Tagrisso (osimertinib), allowing it to be used in Great Britain as a monotherapy for the adjuvant treatment after complete tumour resection in adult patients with stage IB-IIIA non-small cell lung cancer (NSCLC) whose tumours have epidermal growth factor (EGFR) exon 19 deletions or exon 21 (L858R) substitution mutations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
